From Conceptual Design to Commercialization
Whether you have a napkin sketch or a detailed CAD drawing, Natech’s design and engineering team brings your product idea to life.
Novel medical devices require innovative and novel design solutions. Natech engineers work with clients to design and manufacture their medical products. Our team considers the entire product and application when beginning design for manufacturing & assembly (DFMA).
By utilizing our design process, your product can easily transition from product development to next-generation designs as your project scales.
Engineering Services:
Conceptual and Preliminary Product Design
Design for Manufacturing
Design for Assembly
MoldFlow Simulation
Process Optimization
Products are designed for quality and optimal performance. This means a quick and smooth transition from design to manufacturing and final product for you.
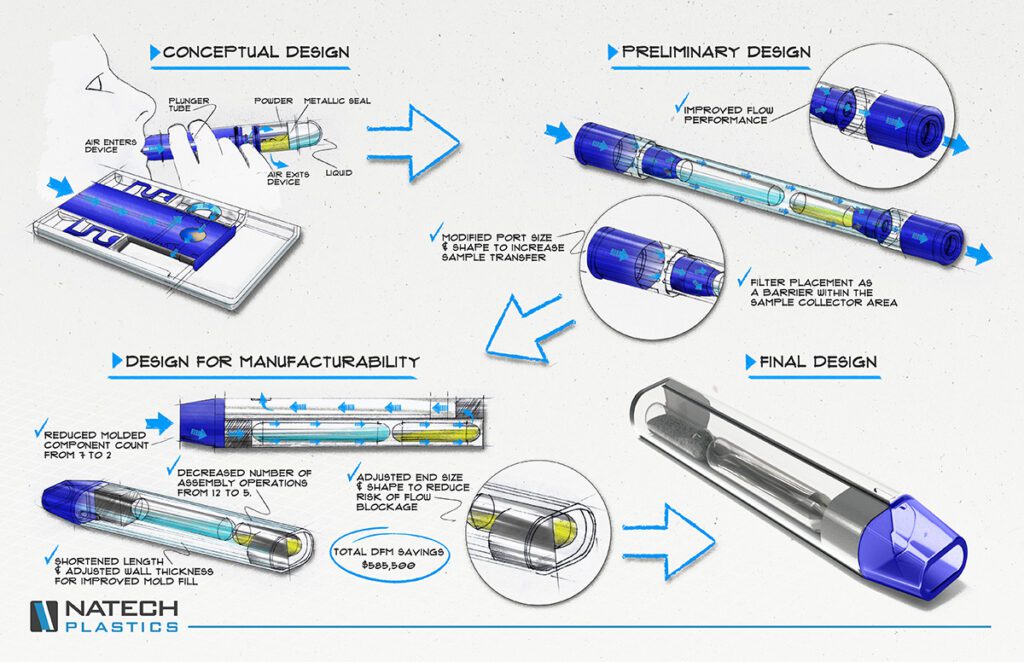
Expert Product Design
Conceptual & Preliminary Design
Customers come to Natech with everything from a napkin sketch to a robust CAD model every day. Our team thrives on guiding clients through the design process and overcoming complex part, material and aesthetic concerns.
Our ability to quickly move from conceptual to final design means a more reliable, higher quality and lower risk product for you.
Design for Manufacturing & Assembly
Our team considers the entire product and application when beginning design for manufacturing & assembly (DFMA).
By incorporating the product’s end use, part functionality, and how it interfaces with other parts or end users, our DFMA gives you confidence that your final design will transition smoothly into manufacture and assembly.

Dedicated Engineering Support
We have developed an intense engineer development program based upon the principles of Quality by Design with a focus on design for manufacturability and design for assembly. Our engineering teams gives clients unparalleled support for their project, regardless of where they are in their development.
Project-Based Support
When you come to Natech, you are assigned to a project engineer for your program. Using our custom project management approach, they work collaboratively with your team to manage the schedule, communicate risks and changes and stay on target and on budget.
Process Optimization
There is no time to waste when launching a new product, and our team is here to ensure an optimal transition into production. That means optimizing the process during design. Our engineers use MoldFlow analysis, automation
potential and scientific processing to optimize your part production from day one.
Quality Engineering
Natech engineers incorporate quality into your product during the design for manufacturing phase. Our in-house DFM expertise and quality engineering team plan ahead to reduce lead times, reduce risks and increase the quality of your final product.
Scientific Validation
The Natech engineers utilize a scientific approach to process validation. This approach reduces risk in the designing, processing and inspecting of the manufacturing process. Scientific validation allows engineers to achieve control of the process and deliver faster and more reliable end results.
Design and Development Resources
-
Five Ways to Eliminate Waste in Cleanroom Manufacturing
Optimizing Cleanroom Manufacturing: Strategies for Efficiency and Cost-Effectiveness Cleanroom manufacturing and assembly play an important role in producing medical devices that demand precise and clean medical devices. To enhance efficiency, reduce costs, and maintain high-quality standards, it is critical to identify key areas where waste can sneak in and where there are practical solutions to…
-
Avoid 3 Engineering Drawing Mistakes with GD&T
Ensure Design Success by Mastering GD&T Principles As a designer specializing in plastic components, crafting precise engineering drawings is paramount for the long-term success of your projects. Over my extensive experience as a CMM metrologist and college professor, I’ve encountered numerous engineering drawings that unintentionally muddle communication and needlessly complicate project requirements. This unfortunate scenario…
-
Beginner’s Guide to Design for Injection Molding
Learn the Basics of Plastic Injection Molding Welcome to the Beginner’s Guide to Design for Injection Molding! This comprehensive guide is designed to provide you with the essential knowledge and insights you need to understand the basics of design for injection molding. Whether you are new to the field or looking to expand your understanding,…
-
How We Used Design for Assembly to Optimize IVD Product
Preventing Leakage and Enhancing User Experience with Design for Assembly An engineer’s core duty includes the seamless integration of design, manufacturing, and utilization. This includes a frequently overlooked yet critical step during the Design for Manufacturing process: Design for Assembly. As organizations expand, sometimes there’s a tendency to separate responsibilities ─ dividing design, manufacturing, and…
-
Mastering the Undercut in Manufacturing
Tradeoffs and Solutions for Effective Undercut Design for Manufacturing Have you ever wondered what challenges keep plastic engineers awake at night? Despite their best efforts, finding the perfect solution without drawbacks can be elusive. In the real world, every solution comes with its own set of risks, forcing them to navigate a delicate balancing act…
-
Reducing Part Variation with Highly Advanced Molds
Solutions for a Complex PCR Molecular Blood Diagnostics Device Application The client: A diagnostics client needed design help, injection molding, and contract manufacturing services for their complex fluidic device. The product: Their PCR molecular blood diagnostics device tests for and identifies multiple viral and bacterial organisms at a time. Problem The client had strict requirements…